CorTec's Stroke Implant Receives FDA Breakthrough Device Designation
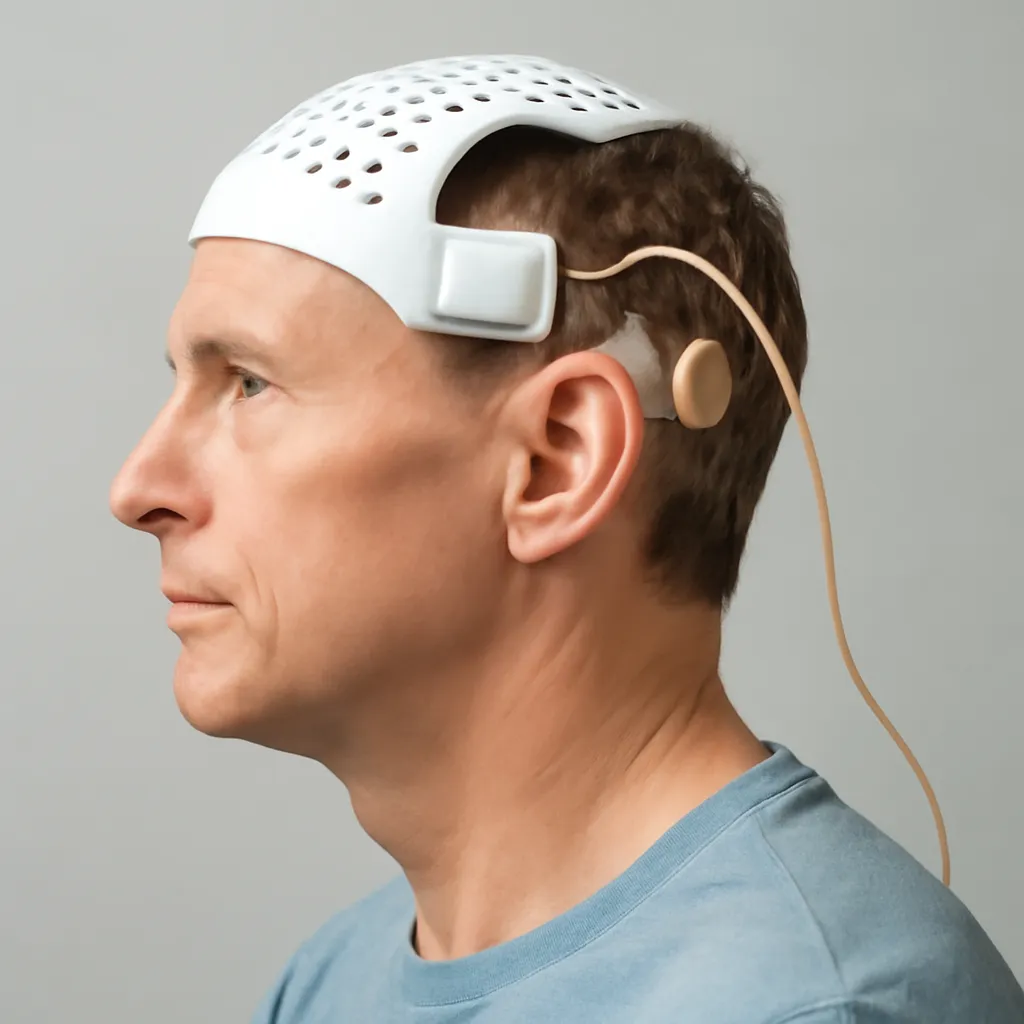
CorTec has announced that its brain-computer interface (BCI) system for stroke rehabilitation has been awarded 'breakthrough device' status by the FDA. This designation is aimed at facilitating the development and expedited FDA review of medical devices that offer significant advantages over existing options. The BCI system is designed to aid in the recovery of stroke patients by enhancing cognitive and motor functions. With this support, CorTec hopes to accelerate the product's availability to those in need. This marks a significant advancement in the field of stroke rehabilitation technology, potentially transforming patient outcomes.
Markets



